Science At Home For Kids
Today, I’m excited to deconstruct the science behind magnets! Magnetism feels like magic! If you’ve ever held two magnets close, they either attract or repel. It feels spooky!
I’m going to show you a cool STEM experiment right at home. This will impress your kids! It’s easy, I promise. Plus, this skill could help you find your way out of the woods. You’ll be a bad…I mean cool, no matter what!
MATERIALS FOR SCIENCE AT HOME : MAGNETS
THREE STEPS TO SCIENCE AT HOME : MAGNETS
Use a big one so the leaf doesn’t hit the sides!
Make sure it’s floating.
Do this for a bout 30 seconds or until they run away, whichever comes first!
The magic WILL happen, be patient.
The needle will point north!
VIDEO OF SCIENCE AT HOME : STEM SCIENCE EXPERIMENT
Happily, dog saliva did not affect the results!
WHAT THE HECK IS GOING ON?
Many of us understand that magnets have two different charges, and that opposite charges attract each other and like charges repel each other. This still does not explain how a magnet works.
Here’s an annoying definition: A magnet is any material or object that produces a magnetic field.
What is with the dictionary anyway? They define words with the word they are supposedly trying to define? That is NOT helpful in any way.
The reason magnets work explained with atoms.
No, really, it is!
ATOMS
Atoms have protons (positive charge) in the nucleus
Atoms also have electrons (negative charge) that orbit around the nucleus.
So what do electrons and protons have to do with magnets?
Moving electrons make a magnetic field.
To make our STEM activity –in the video above– work, we needed to get the electrons to jump from their nice little orbits around their nucleus.
This is hard to do with some atoms. The electrons are snug and tight, paired up and not willing to go anywhere.
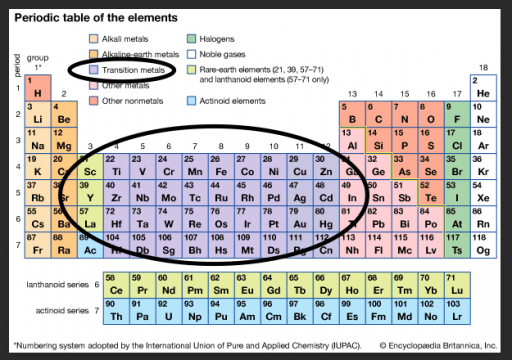
If you look at the periodic table below, the elements on the far right (in the white boxes) have filled orbitals. Those electrons aren’t going anywhere without a fight. This makes the elements very stable. ( Noble Gases)
NOT ALL ATOMS ARE CREATED EQUAL : TRANSITION METALS
Lucky for us, many metals are especially prone to electron conductivity AKA MOVEMENT because they have unpaired electrons in their outer orbitals, spinning around their respective nucleus.
Each orbital has room for a certain number of electrons. (Orbitals are like floors of a building. Once they’re full, a new level must be used)
And once an orbital if full, that makes the atom more stable. (Like our Noble Gases) They are tucked in tightly and tend not to spin off/be attracted to another atom.
If the outer orbital only has a few electrons, they aren’t packed as tightly, have more energy to move, and are more prone to hopping off to find a new home. They’re like teeny tiny explorers.
To make matters more complicated (isn’t that always the case?) with Transition Metals, the third level doesn’t necessarily fill before the fourth begins to fill. WHAT?
Some electrons want their own orbit. (anti-social!)
I know, it stinks when I think I’ve got a rule and it gets broken. (and yes, I’m oversimplifying all this) But you get the general idea!
HOW DO WE GET THE ELECTRONS TO MOVE?
There are three ways to produce an electrical charge.
Friction, induction, and contact.
We’ll just concern ourselves with friction today.
Friction promotes the movement of electrons between two different substances.
On an atomic level, essentially one atom is losing electrons. One will become positively charged (because it lost electrons). The other atom becomes negatively charged. (gained electrons)
When atoms are statically electrified, the electrons actually line up and spin in the same direction.
So what is the point, you ask?
MAGNETIC NORTH
Our planet has a magnetic pole, created by molten lead (LEAD AGAIN) and other metals in the earth’s core.
The molten lead moves as the earth moves! This generates electric currents, which produce magnetic fields!
(My arrows aren’t exactly straight…)
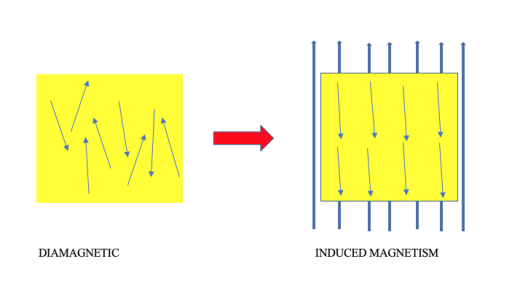
Diamagnetic means not attracted to a magnetic field.
And our metal sewing needle full of lined up electrons (because of the friction) aligns itself with the earth’s strong magnetic field.
This is why compass needles point north, no matter which way you turn the compass.
Field lines are a way to visualize the magnetic field around an object.
MAKING A MAGNET
A lodestone is a piece magnetite, a naturally magnetized metal that attracts iron. The property of magnetism was discovered through lodestones. (FeO4)
Many cultures in the ancient world found lodestones. So discovery of magnetism can’t be pinpointed to any one person.

Lodestone is one of only two minerals that is found naturally magnetized; the other, pyrrhotite, is only weakly magnetic.
Notice the formula for lodestones on the left? They have Fe…we all know which element that is!
How were lodestones formed ?
The most common theory hypothesizes that when iron ore deposits at the surface of the earth are struck by lightning, the electron spin rearranges to match the lightning’s field.
Ancient blacksmiths made magnets in a similar way. The blacksmith on the right is holding a piece of red-hot iron in a north-south direction and hammering it as it cools. The magnetic field of the Earth aligns, leaving the iron a weak magnet.
How did they figure this out?
Spotty wifi connections left them with nothing to do.
MAGNETORECEPTION
Ever wonder how birds migrate so far and find their way? Scientists do, too. The answer is still elusive but it involves MAGNETITE!
Birds, salmon, honeybees and many more animals have magnetite shavings in their brains!
Scientists hypothesize it’s more complex and there may also be a chemical reaction occurring in the eyes of birds.
Birds are cool! They feature in my middle grade fantasy work in progress. To read more about birds, check this post out!

I sure don’t want you to get lost in the woods, but if you do, you’ll be able to find north with this STEM activity. And be super cool.
For now, try it out. Impress your posse. Tell them about electrons jumping and lining up!
Then check back with me and tell me how you did!






















Great post! This was so interesting, I love things like this 🙂
Thanks! Glad you found the post interesting.
Fantastic! I love all of this. How timely, I need some new learning ideas to share with my girls. Thank you.
This will work so go for it! I hope your girls have fun with it.
I love STEM activities! Thanks for sharing this.
I love STEM activities, too. (Not so much taping them, but doing them!) Thanks.
What a cool little experiment and a great way of explaining of magnets work. I think this will be one to share with my niece when she is a little bit older!
Thanks fo sharing 🙂
Aimsy xoxo
Aimsy’s Antics
Thanks for stopping by! Enjoy your niece!
Whoa, this was so informative and cool ! Totally different content and I’m loving it !
Wow, thank you so much! You’ve made my day.
This is so cool! Thanks for sharing, I loved it!
Thanks! I thought it was cool, too. We watched a movie recently with a character finding her way. I had to look into it!
Another great and informative and fun post! Thank you!
Thanks so much! Louie was a huge help.
I love the introduction of magnets for children! Pretty cool how the results turned out with the needle! I am all about science experiments as a kid. I wished there was more back then. Thanks for sharing!
Nancy ♥ exquisitely.me
I love experiments, too. Science is endlessly interesting. Hopefully some kids will think so, too!
I feel like I’m back in school in a science lesson reading your posts! So interesting and informative. I saw this experiment somewhere else a few weeks back – so cool!
I hope mine is better explained! Ha And that being back is school is a good thing. Thanks so much!
Very interesting article! thanks for sharing this. Keep it up!
Will do! Thanks.
My daughter is a massive STEM fan (yay) so I can’t wait to show her this post, I know she’ll want to try this out. I’ve always been fascinated by magnets, and this experiment is a great example of why! Brilliant post, Sue, thank you for sharing 🙂 Lisa
I’m pumped that your daughter loves STEM! I hope she has fun with this experiment! Thanks, Lisa.
Science is cool. Much better use of your time than wanting to play on technology all day.
I agree! My kids are glued to their phones so it was fun to lure them outside and try this experiment! Thanks.
An awesome little experiment anyone could do at home. But I had no idea that that was how magnetism worked. I guess I’d never really thought about it before. I learnt something new today
Hurrah! Thanks for stopping by.
I’m fascinated by the ability of birds/ducks/salmon to find their way back to their birth place, so I appreciated you explanation. Whether it’s magnetite shavings in their brains or a chemical reaction occurring in their eyes, I wish I had that capability since I can get lost in the mall.
HA! Lost in the mall isn’t a bad thing in my book! But yes, I find that ability endlessly interesting, too! I read that humans used to have magnetoreception, but I didn’t dig into it to find actual facts.
This is so interesting, took me back to science at school days!
I love science so I hope taking you back is an OK thing! Thanks.
It is!
Hurrah!
Thank you for sharing this! So cool!!!
Thanks so much!
What a fantastic post 😁 very informative and provides fantastic information to many students 😁
Thanks! I appreciate it!
OMG this is SO COOL!! I love little experiments like this, I can’t wait to test it out!!!
Fantastic! It will definitely work! Thanks so much.
This should be a book! Well done. Perfect for STEM education.
Thanks so much! A book is a fun idea! I’ll look into that.
Ooh I’m trying this, I just need to find a pin…my mum will have one 😀
I did struggle to find other things besides pins, but they are light-weight so make it easy. I hope you do try it!
That is a really cool trick! I’m super intrigued, and want to try it. Thanks for the idea!
You’re welcome! It’s fun AND cool.
An amazing experiment. I used to be a Primary teacher and this would have been amazing to show them!
Lots of love, Helen x
This is high praise, coming from a Primary teacher! Thank you!
I learned so much from this post. Science rules! I often wonder why a compass always point to the north. I wasn’t so keen with directions till I started driving. Now i know, that magnets have atoms. Cool!
Super cool! Thanks.
This was such an interesting post to read. I love anything like this
Glad you liked it! Thanks.
This sounds like such a fun experiment! Thanks for sharing, this is a really interesting post to read! 🙂
melissakacar.blogspot.com
Thank you! I’m happy you stopped by.
Love this. Will be showing the kids tomorrow! Xxx
Fantastic! I’m excited. Let me know how cool they think you are!
Wow! Lots of information about magnets. I always found them interesting and made some cool handmade toys with magnets when my kids were younger.
Magnets are fun, I agree. Handmade toys are the best!
That’s incredible! I have never heard of this before, but it sounds like such a fun experiment! Especially to show children and it’s great that you don’t need any difficult to get a hold of things. Great post xx
Hannah | https://luxuryblush.co.uk/
Thanks! I like to keep it simple, too!
This is such a fun experiment to do, and I love the photo of that sweet little fawn.
Your flip cards also made the neat scientific facts much more engaging. 🙂
I love the flip cards so I’m excited that you do, too! Thanks so much.
I seem to recall them doing something very similar to this at school when they taught us about magnets. Having something like this always makes it seem more fun and easier to remember! x
Sophie
I agree. Hands on helps with learning. And it’s fun! Thanks.
I love reading posts like this they are super interesting, I remember learning about this at school!! Thank you for sharing.
Amber – The Unpredicted Page
Thanks so much! I’m pumped that you liked the post!
I’ve never really done STEM activities before but this looks so much fun and interesting! I love blog posts was where I learn something new! Great post x
I love blog posts when I learn something, too! Thanks so much.
This is such a neat and easy little activity! Great post.
Thanks so much! Easy is key, right?
A cool post loaded with information. Sounds interesting. I am showing this to my son. He’s gonna love it
I’m glad you think so! Let me know what your son says. Thanks so much.
This is such a fun an educational post!
Thank you!
This is so cool!
Thanks!
Wow this was such an interesting and informative post! It’s been ages since I’ve learned anything new about magnets. Thank you for sharing 😊
🌿 Marissa Belle × marsybun.com 🌿
Thanks so much! I’m happy you stopped by.
This seems like such a cool experiment, thanks for sharing!
Tash – http://www.agirlwithview.co.uk
Thanks so much!
this is great
Thanks!