Hand sanitizers or soap? Which is scientifically proven to work best to keep us safer? Soap versus hand sanitizers has become more of an issue than ever!
Let’s look at the science behind soap. And look at the science behind hand sanitizers. We’ll see how each one works and whether or not they destroy viruses and bacteria. Then compare the two. We’ll find our answer. Go science.
I wouldn’t give up my opposable thumbs, but the downside is that our invaluable hands have plenty of nooks and crannies where bacteria and viruses can hide.
Ask any crime scene specialist! Science is knowledge. (And can be used for or against you in a court of law.) Today, we’ll use the science to benefit us!



WHAT ABOUT SURFACES?
HM....
THE SCIENCE OF VIRUSES
When we cough, or especially when we sneeze, tiny droplets from our airways can fly! (gross) And at the time of this post, the controversy over airborne transmission of COVID-19 rages. (It is currently thought that coronavirus transmits via larger droplet transmission. Larger droplets fall quickly, onto surfaces within six feet.)
But let’s go back to virus science so we can scientifically prove if soap or hand sanitizer wins.
These droplets end up on surfaces. They dry out quickly. But the viruses remain active. (Viruses are technically not alive. We won’t go into the why today.)
Human skin is an ideal surface for a virus.
The proteins and fatty acids in the membranes of our dead skin cells interact with the virus.
When you touch a surface with a virus particle, it will stick to your skin and get transferred on to your hands.
HERE’S THE BAD PART:
If you then touch your face, especially your eyes, nostrils, or mouth, you can infect yourself.
And it turns out that most people touch their face once every two to five minutes. Almost half of face touches involve the mouth, nose, and eyes. Those are direct pathways for bacteria and viruses to enter our bodies.
Try your best not to touch your face! Not to bite your nails or cuticles. Pick your nose. Just…DON’T!
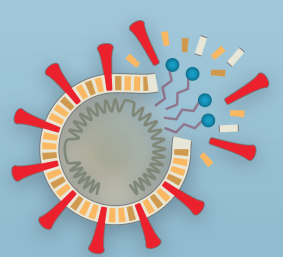
VIRUS CELL MEMBRANES = THE WEAK LINK
Coronaviruses are like many viruses. They have a protected outer surface made of a lipid (fatty) bilayer.
For our purposes, this means they have a head region that is attracted to water (hydrophilic), and a tail region that is repulsed by water. (hydrophobic)
In a water environment (aka you and me), the outer coating forms with the heads out. This helps the virus break through our cell membranes and hijack our cells. (UGH)
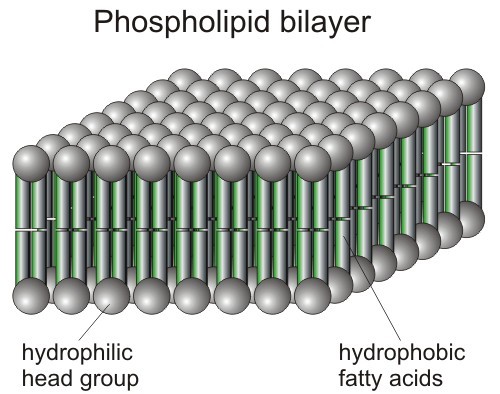
BUT…this lipid bilayer is also the virus’ weakest link!
THE SCIENCE OF SOAP!
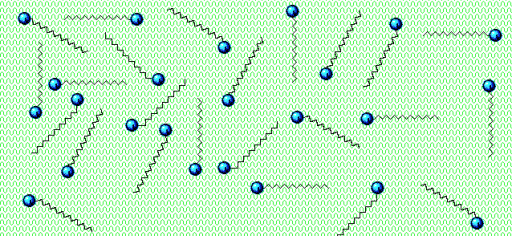
The soap not only loosens the “glue” between the virus and our skin but also the interactions that hold the proteins, lipids, and genetic material in the virus together.
Soap dissolves the fat membrane of the virus – and the virus falls apart! HUZZAH!
But soap doesn’t stop there!
Soap forms particles of their own that surround the virus parts. Apply water, and you’ll wash that bubble with the nasty virus down the drain!
The bubble of soap that circles the broken pieces of virus is called a micelle.
IT TAKES 20 SECONDS
HAVE YOUR KIDS SING THE HAPPY BIRTHDAY SONG
In addition, the rubbing motion while washing with soap and water removes some of the dirty surface skin cells. Who needs those guys, anyway?
THE SCIENCE OF HAND SANITIZERS
Sure, we can read the label of the bottle below but we need to know how to interpret it…which you’ll be able to do because you’re learning about the science of hand sanitizers here!
Have you seen a movie or show when someone gets shot? In westerns or survival movies, the characters sometimes pour alcohol on the wound. The victim winces. The hero takes a swig before expertly beginning his or her patch job.
We laugh or roll our eyes. Or think it’s a good idea, pause the show, and make ourselves a Cosmo.
There is something to be said for that! (The cosmos…pouring alcohol on a wound can cause tissue damage)

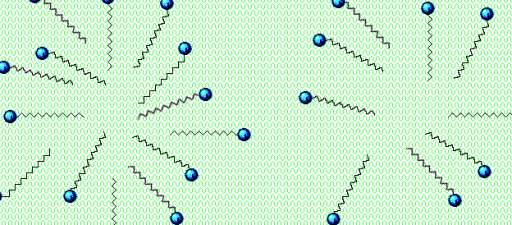
Alcohols perturb the structural arrangement of the membrane lipids. The cell membrane collapses. Scientifically proven!
Alcohol also denatures or disrupts the virus proteins. Viruses have that spikey part, used to penetrate our cells. That is made up of protein.
As the virus breaks down from the alcohol attack, its parts aren’t surrounded in a bubble of soap molecules. (micelle) They crumble. Think of it like taking the nails (or those protein spikes) out of a picture frame. The frame would fall apart.
Do alcohol-based hand rubs destroy the COVID-19 coronavirus?
YES....
Okay, there are a few addendums for hand sanitizers to achieve hero status.
The CDC recommends at least 60% alcohol by volume to effectively knock those viruses on their microscopic butts.
It takes about 30 seconds of rubbing to achieve benefits.
This way you can be more certain that those virus bonds are breaking. (No water to wash it all away…)
Non-alcohol antiseptics rely on benzlkonium chloride instead of alcohol. Don’t be fooled.
Be sure to read the labels!
Don’t purchase anything unless it has at least 60% ethanol or isopropyl alcohol. Not even if it’s on sale.
HAND SANITIZERS OR SOAP? WHICH IS SCIENTIFICALLY PROVEN TO WORK BEST?
In our side-by-side comparison, here are two drawbacks of hand sanitizers….
WILL HAND SANITIZERS EFFECTIVELY RID DIRTY HANDS OF VIRUSES OR BACTERIA?
Sadly, NO
HERE'S ANOTHER CONSIDERATION
SOAP!
Good rule of thumb:
If your hands are dirty, such as before and after eating, using a restroom, playing sports or engaging in any outdoor activities, hand sanitizer won’t be effective.
The sanitizer won’t eliminate the dirt, where the viruses could be hanging out.
We aren’t rubbing hard either, so no surface skin cells are eliminated.
SOAP OR HAND SANITIZERS? FINAL VERDICT
The moment I walk in the door, I wash my hands thoroughly with soap and water. I want you to do this, too!
No, I never sang the happy birthday song before researching this post, but I will from now on. I hope you do as well!
Let’s wash our hands and have a delicious bite to eat!
Which of the three treats would you choose?
Chocolate cake, appetizers, or gelato?


















Perfectly times article, Sue. Love this and have wondered and argued with people. I have always maintained that washing your hands with soap is better, Now I have this article to prove it. Thank you,
Eva
You were right, Eva! I’ve seen conflicting posts online so I decided it was time to research! Thanks!
Much needed article, I’m so here for the Louie content too!
Louie is an entertaining boy! Thanks so much.
I ways knew that soap was better than hand sanitizer, but I didn’t know all the details about why. It’s always nice to learn something new
Now if people disagree with you, you can tell them why you’re right! Thanks.
I use both — with hand sanitizer being a back-up if I can’t wash my hands with soap. Great read!
Thanks so much! Glad you’re fully armed!
Informative and fun post! I carry hand sanitizer with me so that I can disinfect my hands or surfaces when I have to go out. Once I get home, I do lather up with soap and water! Excellent point that soap and water costs much less, too.
Perfect method to keep yourself safer! The money aspect of the equation is often overlooked in these types of discussions. I’m happy that you pointed it out here! Thanks.
We are using soap but recently started using sanitizers as well haha. But this is prove a solid point. Also, science is the best way to determine the truth of everything.
Hand sanitizers have their uses, without question. It’s great to incorporate both. And I agree! Science is a solid way to the truth! Thanks.
Thank you so much for breaking down the science of this complicated subject! This was so informative and entertaining to read! Thanks for sharing! 🙂
melissakacar.blogspot.com
I’m glad you found my post informative and entertaining! Thank you, Melissa.
Something I never knew. Off to buy some soap!
Hurrah! Go soap!
Thanks for sharing this, it is so important to point out that hand sanitiser will not protect you against everything.
Exactly! You’re welcome! Thanks for stopping by.
This was really interesting to read! I always try to keep hand sanitiser with me, especially now!
Tash // A Girl with a View
Good for you to have some along! Thanks.
I used to wonder why I was always getting colds/viruses, then I realised I touch my face almost every 5 minutes, if not more often. I don’t know why, it’s just something I do….trying not to do during this pandemic whilst I’m out and about has been fun and games 🤦♂️😂
Tons of fun, without question! ha
Honestly, I prefer soap over hand sanitizer, but if there isn’t an option, I am down for hand sanitizer too. Thanks for sharing the science behind both!
Nancy ✨ exquisitely.me
I prefer soap, too. Thanks so much!
This is an amazing post, very entertaining too. I use hand sanitizers in cases where I can’t get water but soaps are the best. Great post
Thank you so much! I’m happy that you’re on top of things!
This is a very interesting read. Thank you for sharing this piece!
Thanks so much!
This is such a great and informative post. I learned so much from reading it. Thanks for sharing this. Especially given the circumstances of everything that’s going on right now!
It seemed timely, given the circumstances and conflicting information! Thanks so much, glad you found it useful.
These are so informative, thank you for sharing! I carry hand sanitizer with me everywhere I go and I wash my hands whenever I have access to a sink, I think everyone should get into the habit of doing so too 😊
🌿 Marissa Belle × marsybun.com 🌿
Good for you! I agree, it’s important to develop and stick w/ those important habits. Thanks!
We use both! Thanks for the explanation.
Huzzah! Thanks for stopping by.
Yay for soap! What about “soap nuts”? I bought some at Trader Joe’s and am using them in the laundry. They work great, and there’s no plastic involved. But I wonder if they do the same thing as soap. I won’t be using them on my hands, but I wonder how they work.
Here’s what I found. “Soap berries, also commonly called soap nuts, are the berry-like fruit (related to the lychee fruit) that grows on the Sapindus Mukorrosi tree, native to India and the Himalayas. These berries produce a soap called saponin, which operates as a natural surfactant.”
They sound cool! Thanks for mentioning them!
I have been wondering about this! Glad you posted it. I use hand sanitizer when I am out, but I need to be better about rubbing for 30 seconds…yikes
Me, too! I wasn’t letting it dry completely, either. Now I do! Thanks.
Great column, Sue. Washing it is. And I’ll take that gelato, too.
You’re the first vote on desserts! An excellent choice. Thanks so much, Amy!
This is such an interesting post. I had always wondered if soap would be enough and now I know. In the past I’ve been guilty of touching my face and leaning on my hand. Since Covid started, I’ve made a conscious effort to not do this and the difference in my skin is noticeable. I dread to think what bacteria I was passing onto my skin.
How fantastic that you’ve been successful at stopping an almost unconscious habit! I think if I investigated the answer to that bacteria thought, I’d have to clean far more! ha Thanks!
Very informative and pretty crucial in today’s world! Great post!
Thanks!
This is really interesting, I’ve never thought to compare soap and hand sanitiser. Thank you for sharing.
Thank you for stopping by!
Such an informative post, thank you for breaking the differences down so well!
You’re so welcome! I didn’t discuss toxicity of hand sanitizers because my posts tend to be too long! We definitely need to keep an eye on the kids so they don’t ingest it! Thanks!
Superhero soap! Who knew? A great marketing campaign 🙂 Thanks for all the great info!
Go Soap! Thanks, Eileen!
Great post! I think soap is always the best options, but of course if there’s no soap, hand sanitzer works. Either way, whenever I go out to the groceries and such, I carry a hand sanitizer. But I still feel a bit germophobic and have to wash my hands as soon as I get home. I have a bad habit of touching my face, so this has been a good habit breaker for me.
Grislean | gemlesschosen.com
Sounds as if you’re taking precautions and breaking habits! Huzzah! Thanks so much.
Thank you for this great breakdown and reminder about the importance of washing hands. We’ve been washing our hands a lot more since the pandemic started (like everyone else!) but have started getting lazy and just use hand sanitizer when we’re in a rush. But will make more time to wash with soap!
That sounds just right! Thanks so much.
I knew soap was better, and the 20 seconds thing, but I guess the issue there is more about how well you’re washing your hands. I always use soap at home but keep sanitiser in my bag when I’m on the go x
Sophie
You’ve got this!
Oh wow! This is a super interesting post and a super important thing to talk about!
https://theunpredictedpage.com/first-month-blogging-tasks/
I’m so happy that you think so! Thank you.
We always have soap at home and wash our hands whenever we come in (as well as other times!) but as you say, it’s not practical out and about. Good to know that a combination of the two will work effectively though – great post! Lisa
Thanks, Lisa!
Thanks for this practical advice. More soap, less sanitizer
Perfect! Thanks so much.
This is such a useful post Sue! Definitely highlights the benefits of using both, and keeping our hands clean, something that is so important at the moment. I also didn’t realise how often I touch my face, until I consciously tried to stop!
Thanks for sharing!
Aimsy xoxo
Aimsy’s Antics
I know what you mean, I’m guilty too. A good habit to break! Thanks so much.
I’ve been using both religiously. I always preferred sanitiser though.
Glad you’re using both!
I love using sanitiser and we always have soap around the house. This is a really eye opening article, so thank you for sharing!
Em x
Great that you’re keeping your hands clean using both products. I’m happy that you found my post eye-opening! Thanks so much.
Ooh this is so interesting. I have actually been wondering about this recently!
Great! Glad I could scratch your wondering itch. Thank you.
Very interesting post. Really relevant now for any person on the planet …
Unfortunately, that’s true. Thanks so much.
very well written post more people should read this.
I see many people in the UK
not use a mask after asking me for advice etc as I do H&S.
hopefully next year will be a better year
https://www.wattophotos.co.uk/
Ditto for next year! I’m happy that you’re taking precautions. Thanks so much.
Great post! Thank you a lot! This topic is very important and it’s nice not only to hear the answer but also understand the thing!!!
It is important! You’re most welcome. And thank you!
Super informative! My hands never feel properly clean after using sanitiser, I always prefer to wash them where I can. But it’s good to know that it can still protect you if you can’t get to a tap! Becca x
Agreed! I never feel clean with sanitizer alone either, but yes, at least we know it’s working. Thanks!